How to know that [NiCl4] 2- has tetrahedral geometry whereas [Ni(CN) 4] 2- has square planar geometry - Quora
based VBT Explain the formation of [Ni(CN4)]² - Sarthaks eConnect | Largest Online Education Community
Explain on the basis of valence bond theory that [Ni(CN)4]2− ion with square - Sarthaks eConnect | Largest Online Education Community
Deduce the structures of [NiCl4]^2- and [Ni(CN)4]^2- considering the hybridisation of the metal ion. - Sarthaks eConnect | Largest Online Education Community
![Deduce the structures of [NiCl4]2– and [Ni(CN)4]2– considering the hybridization of the metal ion. Calculate the magnetic moment (spin only) of the species. from Chemistry Coordination Compounds Class 12 Assam Board Deduce the structures of [NiCl4]2– and [Ni(CN)4]2– considering the hybridization of the metal ion. Calculate the magnetic moment (spin only) of the species. from Chemistry Coordination Compounds Class 12 Assam Board](https://www.zigya.com/application/uploads/images/chen12070397-a_5714962c868f8.png?t=1460966959418)
Deduce the structures of [NiCl4]2– and [Ni(CN)4]2– considering the hybridization of the metal ion. Calculate the magnetic moment (spin only) of the species. from Chemistry Coordination Compounds Class 12 Assam Board
How to know that [NiCl4] 2- has tetrahedral geometry whereas [Ni(CN) 4] 2- has square planar geometry - Quora
![inorganic chemistry - Why is [PdCl4]2- square planar whereas [NiCl4]2- is tetrahedral? - Chemistry Stack Exchange inorganic chemistry - Why is [PdCl4]2- square planar whereas [NiCl4]2- is tetrahedral? - Chemistry Stack Exchange](https://i.stack.imgur.com/xHv3g.png)
inorganic chemistry - Why is [PdCl4]2- square planar whereas [NiCl4]2- is tetrahedral? - Chemistry Stack Exchange
![Account for the following on the basis of valence bond theory. (i) [Ni(CN)4 ]^(2-) is diamagnetic and square planar. (ii) [NiC4]^(2-) is paramagnetic and tetrahedral. Account for the following on the basis of valence bond theory. (i) [Ni(CN)4 ]^(2-) is diamagnetic and square planar. (ii) [NiC4]^(2-) is paramagnetic and tetrahedral.](https://d10lpgp6xz60nq.cloudfront.net/physics_images/ARH_CHSE_ODI_13Y_SP_CHE_XII_C13_E02_018_S01.png)
Account for the following on the basis of valence bond theory. (i) [Ni(CN)4 ]^(2-) is diamagnetic and square planar. (ii) [NiC4]^(2-) is paramagnetic and tetrahedral.
![Deduce the structures of [NiCl4]2– and [Ni(CN)4]2– considering the hybridization of the metal ion. Calculate the magnetic moment (spin only) of the species. from Chemistry Coordination Compounds Class 12 Haryana Board - Deduce the structures of [NiCl4]2– and [Ni(CN)4]2– considering the hybridization of the metal ion. Calculate the magnetic moment (spin only) of the species. from Chemistry Coordination Compounds Class 12 Haryana Board -](https://www.zigya.com/application/uploads/images/chen12070397_571494e890adc.png)
Deduce the structures of [NiCl4]2– and [Ni(CN)4]2– considering the hybridization of the metal ion. Calculate the magnetic moment (spin only) of the species. from Chemistry Coordination Compounds Class 12 Haryana Board -
![Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson](https://i.pinimg.com/originals/a2/20/10/a22010863c43298057a81f0eb09dd692.jpg)
Diamagnetic - Paramagnetic - Ni(CO)4, [Ni(CN)4]2- and [NiCl4]2- IIT JEE ... | Chemistry lessons, Chemistry, Lesson
![For the complex [NiCl4]2-, write (i) The IUPAC name. (ii) The hybridization type. (iii) The shape of the complex. (Atomic no. of Ni = 28) from Chemistry Coordination Compounds Class 12 CBSE For the complex [NiCl4]2-, write (i) The IUPAC name. (ii) The hybridization type. (iii) The shape of the complex. (Atomic no. of Ni = 28) from Chemistry Coordination Compounds Class 12 CBSE](https://www.zigya.com/application/zrc/images/qvar/CHEN12112201.png)

![Hybridization and geometry of [Ni(CN)4]^2 - are: Hybridization and geometry of [Ni(CN)4]^2 - are:](https://haygot.s3.amazonaws.com/questions/1453910_739662_ans_1b88a50e6b3142878d714df46d9472b7.png)
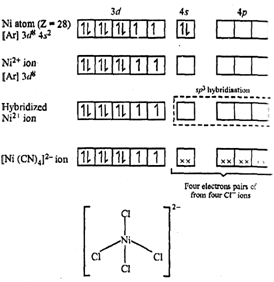
![The geometry of [Ni(CN)4]^2 - and [NiCl4]^2 - ions are : The geometry of [Ni(CN)4]^2 - and [NiCl4]^2 - ions are :](https://haygot.s3.amazonaws.com/questions/345819_212776_ans_5a2a220d536a4adebbfad43e7ddf74b5.png)
![Hybridization and geometry of [Ni(CN)4]^2 - are: Hybridization and geometry of [Ni(CN)4]^2 - are:](https://haygot.s3.amazonaws.com/questions/623286_597576_ans_83f06d1a64a9465c875f91d8efb8e27d.png)

![Hybridization and geometry of [Ni(CN)4]^2 - are: Hybridization and geometry of [Ni(CN)4]^2 - are:](https://haygot.s3.amazonaws.com/questions/13923_22300_ans_70a24a98620f4cf0a7c6cf4edff26bcf.png)

![Ni(CO)4, [Ni(CN)4]2-, [NiCl4]2-Structure-Hybridization-VBT-IIT JEE NEET SAT NCERT CBSE - YouTube Ni(CO)4, [Ni(CN)4]2-, [NiCl4]2-Structure-Hybridization-VBT-IIT JEE NEET SAT NCERT CBSE - YouTube](https://i.ytimg.com/vi/r_C4yyTUSjM/maxresdefault.jpg)
![Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure -Parmagnetic-Diamagnetic-Examples-dsp2 Hybridization-Ni(CO)4 | [Ni(CN)4]2-| [Ni(Cl)4]2- | Structure -Parmagnetic-Diamagnetic-Examples-dsp2](http://www.adichemistry.com/jee/qb/coordination-chemistry/1/q1-1.png)