![FTIR spectra of a Ni(Sal)2, b [Cr(en)3]Cl3.3H2O, and c NiCr2O4 (sample 2) | Download Scientific Diagram FTIR spectra of a Ni(Sal)2, b [Cr(en)3]Cl3.3H2O, and c NiCr2O4 (sample 2) | Download Scientific Diagram](https://www.researchgate.net/publication/313686161/figure/fig1/AS:941596942024714@1601505613109/FTIR-spectra-of-a-NiSal2-b-Cren3Cl33H2O-and-c-NiCr2O4-sample-2.gif)
FTIR spectra of a Ni(Sal)2, b [Cr(en)3]Cl3.3H2O, and c NiCr2O4 (sample 2) | Download Scientific Diagram

The calculated molar absorbance spectra for nickel species (Ni 2+ ,... | Download Scientific Diagram
![Scanning electron microscope (SEM) images of (a) [Fe(L)]Cl3 and (b)... | Download Scientific Diagram Scanning electron microscope (SEM) images of (a) [Fe(L)]Cl3 and (b)... | Download Scientific Diagram](https://www.researchgate.net/publication/356820649/figure/fig4/AS:1117440197033984@1643429912414/Scanning-electron-microscope-SEM-images-of-a-FeLCl3-and-b-NiLCl2-complexes.png)
Scanning electron microscope (SEM) images of (a) [Fe(L)]Cl3 and (b)... | Download Scientific Diagram

SOLVED: How many Ampers (A) are needed to deposit 0.4685 g of Ni metal from an aqueous solution of NiCl3, in 495 seconds?

Ions Atoms that lose electrons (negative) have more positive charge than negative charge so they are positive ions. Atoms that gain electrons (negative) - ppt download
Among (i) [Co(NH3)6]Cl3, (ii) [Ni(NH3)6]Cl2, (iii) [Cr(H2O)6]Cl3, (iv) [Fe(H2O)6]Cl2 the complex which is diamagnetic is - Sarthaks eConnect | Largest Online Education Community
![Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium](https://miro.medium.com/v2/resize:fit:1090/0*rkWRh7JsXBvtOfdK.jpg)
Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
![Ni(NH3)6]Cl2 paramagnetic but [Co(NH3)6]Cl3 is diamagnetic - CHEMSOLVE.NET | Electron configuration, Crystal field theory, Coordination number Ni(NH3)6]Cl2 paramagnetic but [Co(NH3)6]Cl3 is diamagnetic - CHEMSOLVE.NET | Electron configuration, Crystal field theory, Coordination number](https://i.pinimg.com/736x/cb/55/b9/cb55b933ee9ec4d59fdf9d226cf3a51a.jpg)
Ni(NH3)6]Cl2 paramagnetic but [Co(NH3)6]Cl3 is diamagnetic - CHEMSOLVE.NET | Electron configuration, Crystal field theory, Coordination number
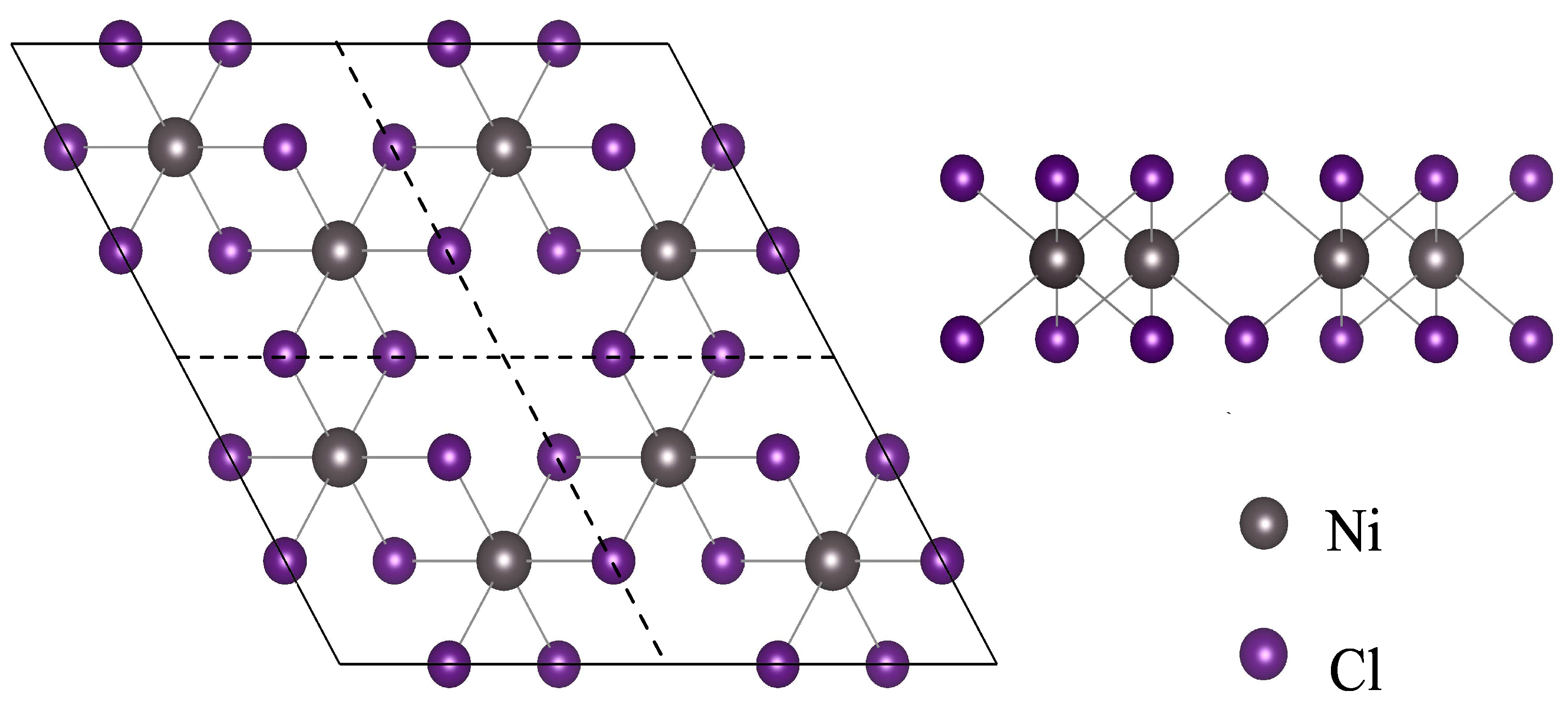
Nanomaterials | Free Full-Text | Thermoelectric Properties of NiCl3 Monolayer: A First-Principles-Based Transport Study
![Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium](https://miro.medium.com/v2/resize:fit:1280/0*8wwL8Ru43LyLIIL8.png)
Why Is [Ni(NH3)6]Cl2 Paramagnetic But [Co(NH3)6]Cl3 Is Diamagnetic ? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
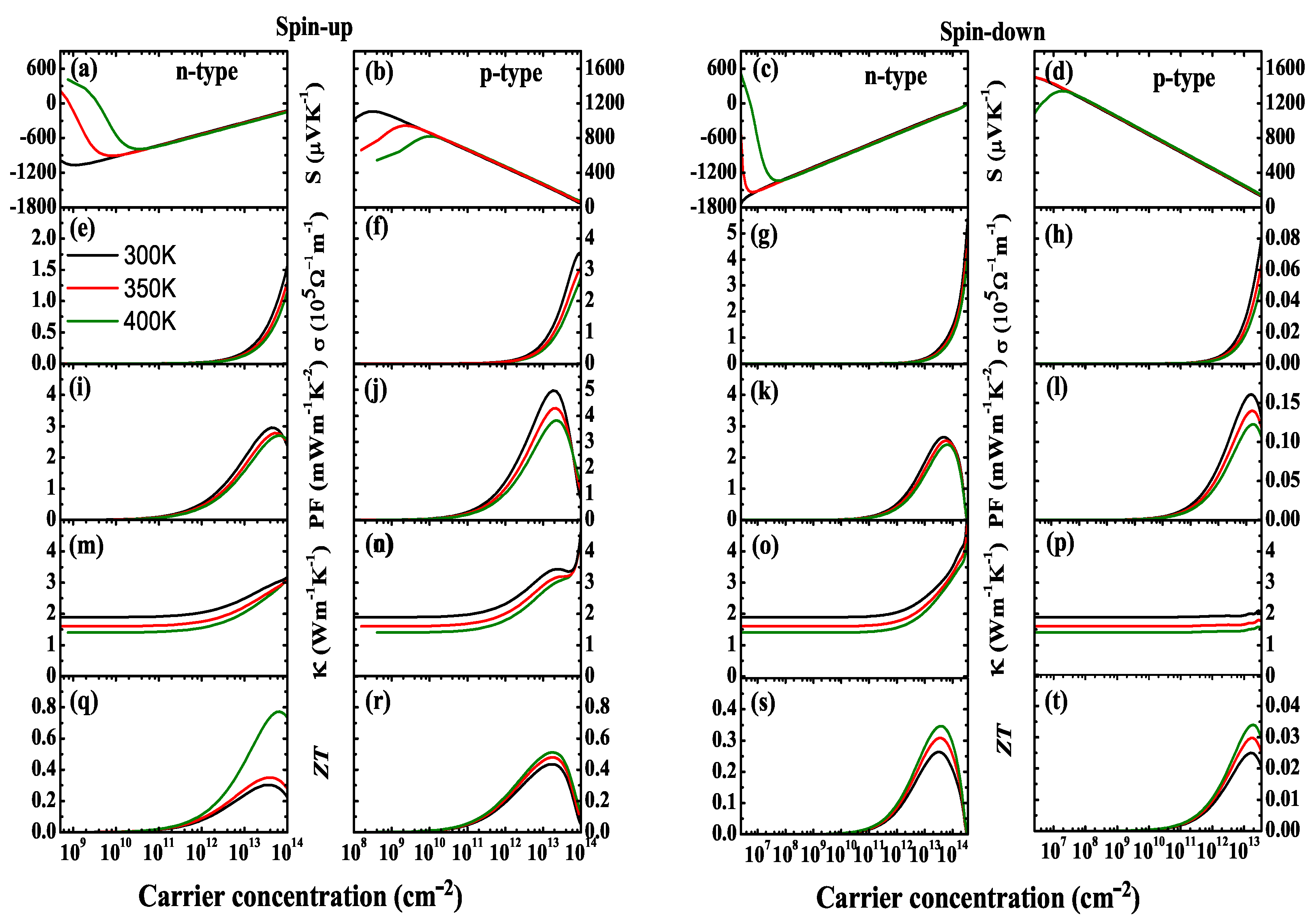
![PDF] NiCl3 Monolayer: Dirac Spin-Gapless Semiconductor and Chern Insulator | Semantic Scholar PDF] NiCl3 Monolayer: Dirac Spin-Gapless Semiconductor and Chern Insulator | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/2c823e3baa5056e082b087e7244c0d89871a55a8/4-Figure1-1.png)
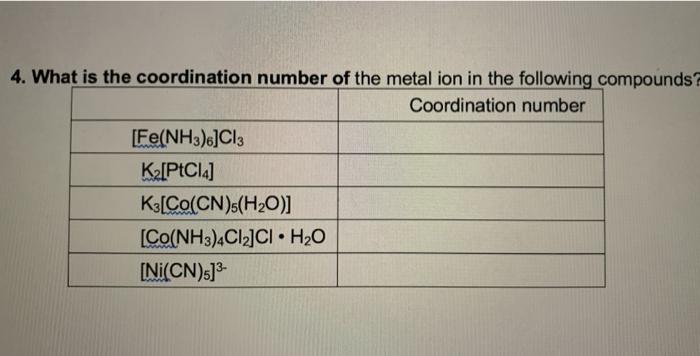







![Solved 5. Name the following compounds. a) [Cr(NH3)6]Cl3 b) | Chegg.com Solved 5. Name the following compounds. a) [Cr(NH3)6]Cl3 b) | Chegg.com](https://media.cheggcdn.com/media/a6d/s918x302/a6df084b-878c-469e-989e-bb7aab4ee9c3/image.png)

